crystal field theory
Crystal field theory (CFT) is a model that describes the breaking of degeneracies of electronic orbital states, usually d or f orbitals, due to a static electric field produced by a surrounding charge distribution (anion neighbors). This theory has been used to describe various spectroscopies of transition metal coordination complexes, in particular optical spectra (colours). CFT successfully accounts for some magnetic properties, colours, hydration enthalpies, andspinel structures of transition metal complexes, but it does not attempt to describe bonding. CFT was developed by physicists Hans Bethe and John Hasbrouck van Vleck[1] in the 1930s. CFT was subsequently combined with molecular orbital theory to form the more realistic and complex ligand field theory (LFT), which delivers insight into the process of chemical bonding in transition metal complexes.
Contents[hide] |
[edit]Overview of crystal field theory analysis
According to CFT, the interaction between a transition metal and ligands arises from the attraction between the positively charged metal cation and negative charge on the non-bonding electrons of the ligand. The theory is developed by considering energy changes of the five degenerate d-orbitals upon being surrounded by an array of point charges consisting of the ligands. As a ligand approaches the metal ion, the electrons from the ligand will be closer to some of thed-orbitals and farther away from others causing a loss of degeneracy. The electrons in the d-orbitals and those in the ligand repel each other due to repulsion between like charges. Thus the d-electrons closer to the ligands will have a higher energy than those further away which results in the d-orbitals splitting in energy. This splitting is affected by the following factors:
- the nature of the metal ion.
- the metal's oxidation state. A higher oxidation state leads to a larger splitting.
- the arrangement of the ligands around the metal ion.
- the nature of the ligands surrounding the metal ion. The stronger the effect of the ligands then the greater the difference between the high and low energy d groups.
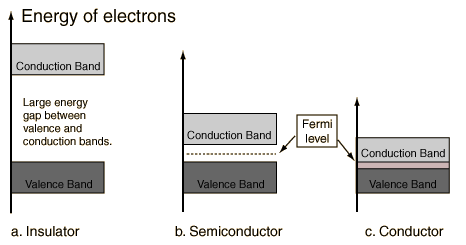
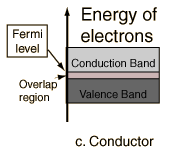
The most common type of complex is octahedral; here six ligands form an octahedron around the metal ion. In octahedral symmetry the d-orbitals split into two sets with an energy difference, Δoct (the crystal-field splitting parameter) where the dxy, dxz and dyz orbitals will be lower in energy than the dz2 and dx2-y2, which will have higher energy, because the former group is farther from the ligands than the latter and therefore experience less repulsion. The three lower-energy orbitals are collectively referred to as t2g, and the two higher-energy orbitals as eg. (These labels are based on the theory of molecular symmetry). Typical orbital energy diagrams are given below in the section High-spin and low-spin.
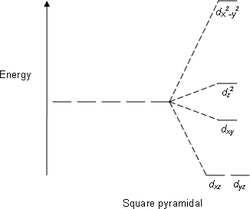
Tetrahedral complexes are the second most common type; here four ligands form a tetrahedron around the metal ion. In a tetrahedral crystal field splitting the d-orbitals again split into two groups, with an energy difference of Δtet where the lower energy orbitals will be dz2 and dx2-y2, and the higher energy orbitals will be dxy, dxz and dyz - opposite to the octahedral case. Furthermore, since the ligand electrons in tetrahedral symmetry are not oriented directly towards thed-orbitals, the energy splitting will be lower than in the octahedral case. Square planar and other complex geometries can also be described by CFT.
The size of the gap Δ between the two or more sets of orbitals depends on several factors, including the ligands and geometry of the complex. Some ligands always produce a small value of Δ, while others always give a large splitting. The reasons behind this can be explained by ligand field theory. The spectrochemical series is an empirically-derived list of ligands ordered by the size of the splitting Δ that they produce (small Δ to large Δ; see also this table):
I− < Br− < S2− < SCN− < Cl− < NO3− < N3− < F− < OH− < C2O42− < H2O < NCS− < CH3CN < py < NH3 < en < 2,2'-bipyridine < phen < NO2− < PPh3 < CN− < CO
It is useful to note that the ligands producing the most splitting are those that can engage in metal to ligand back-bonding.
The oxidation state of the metal also contributes to the size of Δ between the high and low energy levels. As the oxidation state increases for a given metal, the magnitude of Δ increases. A V3+ complex will have a larger Δ than a V2+complex for a given set of ligands, as the difference in charge density allows the ligands to be closer to a V3+ ion than to a V2+ ion. The smaller distance between the ligand and the metal ion results in a larger Δ, because the ligand and metal electrons are closer together and therefore repel more.
[edit]High-spin and low-spin
Main article: Spin states (d electrons)
See also: Magnetochemistry
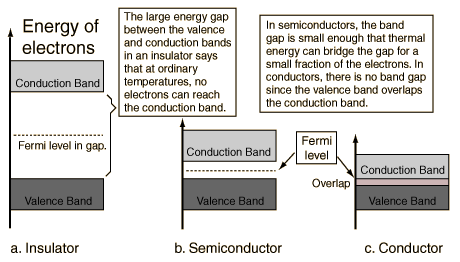
Ligands which cause a large splitting Δ of the d-orbitals are referred to as strong-field ligands, such as CN− and CO from the spectrochemical series. In complexes with these ligands, it is unfavourable to put electrons into the high energy orbitals. Therefore, the lower energy orbitals are completely filled before population of the upper sets starts according to the Aufbau principle. Complexes such as this are called "low spin". For example, NO2− is a strong-field ligand and produces a large Δ. The octahedral ion [Fe(NO2)6]3−, which has 5 d-electrons, would have the octahedral splitting diagram shown at right with all five electrons in the t2g level.
Conversely, ligands (like I− and Br−) which cause a small splitting Δ of the d-orbitals are referred to as weak-field ligands. In this case, it is easier to put electrons into the higher energy set of orbitals than it is to put two into the same low-energy orbital, because two electrons in the same orbital repel each other. So, one electron is put into each of the five d-orbitals before any pairing occurs in accord with Hund's rule and "high spin" complexes are formed. For example, Br− is a weak-field ligand and produces a small Δoct. So, the ion [FeBr6]3−, again with five d-electrons, would have an octahedral splitting diagram where all five orbitals are singly occupied.
In order for low spin splitting to occur, the energy cost of placing an electron into an already singly occupied orbital must be less than the cost of placing the additional electron into an egorbital at an energy cost of Δ. As noted above, eg refers to the dz2 and dx2-y2 which are higher in energy than the t2g in octahedral complexes. If the energy required to pair two electrons is greater than the energy cost of placing an electron in an eg, Δ, high spin splitting occurs.
The crystal field splitting energy for tetrahedral metal complexes (four ligands) is referred to as Δtet, and is roughly equal to 4/9Δoct (for the same metal and same ligands). Therefore, the energy required to pair two electrons is typically higher than the energy required for placing electrons in the higher energy orbitals. Thus, tetrahedral complexes are usually high-spin.
The use of these splitting diagrams can aid in the prediction of the magnetic properties of coordination compounds. A compound that has unpaired electrons in its splitting diagram will be paramagnetic and will be attracted by magnetic fields, while a compound that lacks unpaired electrons in its splitting diagram will be diamagnetic and will be weakly repelled by a magnetic field.
[edit]Crystal field stabilization energy
The crystal field stabilization energy (CFSE) is the stability that results from placing a transition metal ion in the crystal field generated by a set of ligands. It arises due to the fact that when the d-orbitals are split in a ligand field (as described above), some of them become lower in energy than before with respect to a spherical field known as the barycenter in which all five d-orbitals are degenerate. For example, in an octahedral case, the t2g set becomes lower in energy than the orbitals in the barycenter. As a result of this, if there are any electrons occupying these orbitals, the metal ion is more stable in the ligand field relative to the barycenter by an amount known as the CFSE. Conversely, theeg orbitals (in the octahedral case) are higher in energy than in the barycenter, so putting electrons in these reduces the amount of CFSE.
If the splitting of the d-orbitals in an octahedral field is Δoct, the three t2g orbitals are stabilized relative to the barycenter by 2/5 Δoct, and the egorbitals are destabilized by 3/5 Δoct. As examples, consider the two d5 configurations shown further up the page. The low-spin (top) example has five electrons in the t2g orbitals, so the total CFSE is 5 x 2/5 Δoct = 2Δoct. In the high-spin (lower) example, the CFSE is (3 x 2/5 Δoct) - (2 x 3/5Δoct) = 0 - in this case, the stabilization generated by the electrons in the lower orbitals is canceled out by the destabilizing effect of the electrons in the upper orbitals.
Crystal Field stabilization is applicable to transition-metal complexes of all geometries. Indeed, the reason that many d8 complexes are square-planar is the very large amount of crystal field stabilization that this geometry produces with this number of electrons.
[edit]Explaining the colors of transition metal complexes
The bright colors exhibited by many coordination compounds can be explained by Crystal Field Theory. If the d-orbitals of such a complex have been split into two sets as described above, when the molecule absorbs a photon of visible light one or more electrons may momentarily jump from the lower energy d-orbitals to the higher energy ones to transiently create an excited state atom. The difference in energy between the atom in the ground state and in the excited state is equal to the energy of the absorbed photon, and related inversely to the wavelength of the light. Because only certain wavelengths (λ) of light are absorbed - those matching exactly the energy difference - the compounds appears the appropriate complementary color.
As explained above, because different ligands generate crystal fields of different strengths, different colors can be seen. For a given metal ion, weaker field ligands create a complex with a smaller Δ, which will absorb light of longer λ and thus lower frequency ν. Conversely, stronger field ligands create a larger Δ, absorb light of shorter λ, and thus higher ν. It is, though, rarely the case that the energy of the photon absorbed corresponds exactly to the size of the gap Δ; there are other things (such as electron-electron repulsion and Jahn-Teller effects) that also affect the energy difference between the ground and excited states.
[edit]Which colors are exhibited?
This color wheel demonstrates which color a compound will appear if it only has one absorption in the visible spectrum. For example, if the compound absorbs red light, it will appear green.
λ absorbed versus color observed
400 nm Violet absorbed, Green-yellow observed (λ 560 nm)
450 nm Blue absorbed, Yellow observed (λ 600 nm)
490 nm Blue-green absorbed, Red observed (λ 620 nm)
570 nm Yellow-green absorbed, Violet observed (λ 410 nm)
580 nm Yellow absorbed, Dark blue observed (λ 430 nm)
600 nm Orange absorbed, Blue observed (λ 450 nm)
650 nm Red absorbed, Green observed (λ 520 nm)
400 nm Violet absorbed, Green-yellow observed (λ 560 nm)
450 nm Blue absorbed, Yellow observed (λ 600 nm)
490 nm Blue-green absorbed, Red observed (λ 620 nm)
570 nm Yellow-green absorbed, Violet observed (λ 410 nm)
580 nm Yellow absorbed, Dark blue observed (λ 430 nm)
600 nm Orange absorbed, Blue observed (λ 450 nm)
650 nm Red absorbed, Green observed (λ 520 nm)
[edit]Crystal field splitting diagrams
| Octahedral | Pentagonal bipyramidal | Square antiprismatic |
|---|---|---|
| Square planar | Square pyramidal | Tetrahedral |
| Trigonal bipyramidal | ||